Professor Wenhao Sun displays dolomite from his personal rock collection. Sun studies the crystal growth of metals from a materials science perspective. By understanding how atoms come together to form natural minerals, he believes we can uncover the basic mechanisms of crystal growth, which can be used to make functional materials more quickly and efficiently. Credit: Marcin Szczybanski, Senior Multimedia Storyteller, Michigan Engineering.
To create mountains of dolomite, a common mineral, it must be melted periodically. This seemingly contradictory concept can help make new products flawless Semiconductors And more.
For two centuries, scientists have failed to produce a common mineral in the laboratory under conditions believed to have formed naturally. Now, a team of researchers from the University of Michigan and Hokkaido University In Sapporo, Japan has finally achieved just that, thanks to a new theory developed through atomic simulations.
Their success solves a long-standing geological mystery called the “Dolomite Problem.” Dolomite – a major mineral found in the Dolomite Mountains in Italy, Niagara Falls, and Hoodoo in Utah – is abundant in rocks Older than 100 million yearsHowever, it is almost absent in young formations.

Wenhao Sun, Dow Assistant Professor of Materials Science and Engineering at the University of Michigan, and Junsu Kim, a materials science and engineering doctoral student in Professor Sun's research group, show dolomite rocks from their lab's collection. The two scientists have developed a theory that could finally explain a two-century-old mystery about the abundance of dolomite on Earth. Credit: Marcin Szczybanski, Senior Multimedia Storyteller, Michigan Engineering.
The importance of understanding dolomite growth
“If we understand how dolomite grows in nature, we may learn new strategies to enhance the crystal growth of modern technological materials,” Wenhao Sun, a professor of materials science and engineering at Dow University and corresponding author on the paper, said recently. Published in Sciences.
The secret to finally growing dolomite in the laboratory was removing defects in the mineral structure as it grew. When minerals form in water, atoms are usually deposited neatly at the edge of the growing crystalline surface. However, the growth edge of dolomite consists of alternating rows of calcium and magnesium. In water, calcium and magnesium attach randomly to the growing dolomite crystal, often settling in the wrong place and creating defects that prevent additional layers of dolomite from forming. This disturbance slows the growth of the dolomite to a crawl, meaning it would take 10 million years to make just one layer of ordered dolomite.
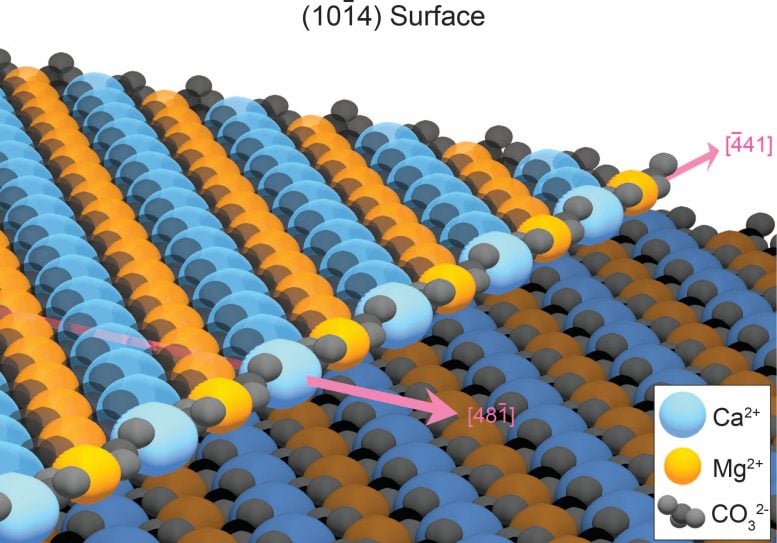
Dolomite crystal edge structure. Rows of magnesium (orange balls) alternate with rows of calcium (blue balls), interspersed with carbonates (black structures). Pink arrows show crystal growth directions. Calcium and magnesium often bind to the growth edge incorrectly, stopping dolomite growth. Image source: Junsu Kim, PhD student in Materials Science and Engineering, University of Michigan.
Fortunately, these defects are not fixed in place. Because disordered atoms are less stable than atoms in the correct position, they are the first to dissolve when the metal is washed with water. Repeatedly washing away these faults—for example, with rain or tidal cycles—allows the dolomite layer to form in just a matter of years. Over geological time, dolomite mountains can accumulate.
Advanced simulation techniques
To accurately simulate dolomite growth, the researchers needed to calculate how strongly or weakly the atoms were attached to the surface of the existing dolomite. More accurate simulations require the energy of each interaction between electrons and atoms in the growing crystal. Such exhaustive calculations typically require massive amounts of computing power, but software developed at the University of Maryland's Center for Predictive Structural Materials Science (PRISMS) has provided a shortcut.
“Our software calculates the energy of some atomic arrangements, and then extrapolates them to predict the energies of other arrangements based on the symmetry of the crystal structure,” said Brian Buchala, one of the program's lead developers and an associate research scientist in the University of Maryland department. Materials science and engineering.
This shortcut made it possible to simulate dolomite growth over geological timescales.

Dolomite is a mineral so common in ancient rocks that it forms mountains such as the mountain range of the same name in northern Italy. But dolomite is rare in younger rocks and cannot be made in a laboratory under the conditions in which it formed naturally. A new theory has helped scientists grow the mineral in the laboratory at normal temperature and pressure for the first time, and could help explain the scarcity of dolomite in younger rocks. Image source: Francesca.z73 via Wikimedia Commons.
“Each atomic step typically takes more than 5,000 CPU hours on a supercomputer. Now, we can do the same calculation in 2 milliseconds on a desktop,” said Junsu Kim, a PhD student in materials science and engineering and first author of the study.
Practical application and theory testing
The few areas where dolomite forms today are intermittently flooded and later dry up, which agrees well with Sun and Kim's theory. But such evidence alone was not sufficient to be completely convincing. Enter Yuki Kimura, a professor of materials science from Hokkaido University, and Tomoya Yamazaki, a postdoctoral researcher in Kimura's lab. They tested the new theory using transmission electron microscopes.
“Electron microscopes typically only use electron beams to image samples,” Kimura said. “However, the beam can also split the water, making… sour Which can cause the crystals to dissolve. This is usually a bad thing for photography, but in this case, the decomposition is exactly what we wanted.
After placing a small dolomite crystal in a solution of calcium and magnesium, Kimura and Yamazaki gently pulsed the electron beam 4,000 times over two hours, removing the defects. After the pulses, the dolomite was seen growing by about 100 nanometers, about 250,000 times smaller than an inch. Although these were only 300 layers of dolomite, no more than five layers of dolomite had ever been grown in a laboratory before.
Lessons learned from the dolomite problem could help engineers manufacture higher-quality materials for semiconductors, solar panels, batteries and other technology.
“In the past, crystal growers who wanted to make flawless materials would try to grow them very slowly,” Sun said. “Our theory shows that you can grow defect-free materials quickly, if you dissolve the defects periodically during growth.”
Reference: “Melting Enables Growth of Dolomite Crystals at Near Ambient Conditions” by Junsu Kim, Yuki Kimura, Brian Buchala, Tomoya Yamazaki, Udo Becker, and Wenhao Sun, November 23, 2023, Sciences.
doi: 10.1126/science.adi3690
The research was funded by a New Doctoral Investigator Grant from the American Chemical Society PRF, the U.S. Department of Energy, and the Japan Society for the Promotion of Science.

“Beer aficionado. Gamer. Alcohol fanatic. Evil food trailblazer. Avid bacon maven.”
